|
Chemguide: Core Chemistry 14 - 16 Changes of state between solids, liquids and gases This page looks at what happens to the particles in solids, liquids and gases during changes of state. The purpose of this page is to encourage you to think about simple everyday things in terms of particles - their energy, their attractions and their movement. Changes of state between solid and liquid Melting Remember that particles in a solid are fixed in position and although they can't move around, they are vibrating. They are held together in the solid by forces of attraction between the various particles. When you heat a solid, energy is transferred to the particles and makes them vibrate more strongly. Eventually, they are vibrating so much that the attractive forces are no longer strong enough to hold them together as a solid. So the solid melts.
It is important to realise that although the forces of attraction between the particles in the solid have been loosened, they haven't been fully broken. If they were, the particles would all fly apart to give a gas. How much you have to heat a solid before it melts depends on the strength of the forces between the particles. For example, sulfur melts at 113°C while tungsten melts at 3410°C. The forces holding tungsten particles together are obviously very much stronger than those holding the sulfur particles together. Freezing Freezing reverses this. In a liquid, the particles have enough room to move around, but they are still close together, and still have attractive forces between them. But at the temperature of the liquid, those forces aren't strong enough to overcome the energy of the moving particles and trap them into a solid. As you cool a liquid, removing energy from it, the movement of the particles gets slower. At some point they get slow enough that the attractive forces are strong enough to trap them and lock them into the solid structure. The liquid freezes. Changes of state between liquid and gas Boiling If you heat a liquid strongly enough, eventually the particles will be moving around with enough energy that they overcome the forces holding them together in the liquid. When that happens, the particles fly off as a gas. Condensing The attractions between particles in a gas aren't strong enough for the particles to stick together as a liquid if they collide. But there are attractive forces. If you lower the temperature enough and remove enough energy from the particles, every gas will sooner or later condense to a liquid. (Or even a solid! Carbon dioxide, for example, at ordinary pressures doesn't form a liquid on cooling - it goes straight to the solid.) Even helium, the substance with the lowest boiling point, condenses to a liquid at -269°C at ordinary pressure: just 4°C above absolute zero. The attractions between helium particles are very very small. Evaporation It is important that you don't confuse evaporation and boiling. They happen entirely differently. 
The photo shows water boiling in a glass kettle. It is obvious if you look at it that boiling is a very violent process. The water particles are breaking away from each other to form a gas (steam) throughout the liquid. The large increase in volume when water converts to steam accounts for all the bubbles and jumping-around of the liquid. Evaporation is completely different.
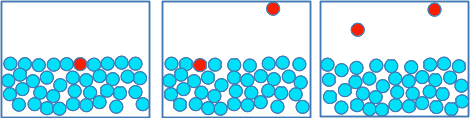
A puddle of water just sits there quietly, and eventually (faster if it is sunny or warm) disappears. In this case, the water particles are breaking away only from the surface. So how do some of the surface particles manage to break away from the attractions of the other water particles underneath them? An aside on temperature and energy Temperature is a measure of the average energy due to the movements of the particles in a substance. That means that if you have two substances at the same temperature, the average energy due to movement of the particles in both substances is the same. If you add more energy, the temperature increases because the average energy due to movement of the particles has increased. But this is an average energy we are talking about. Within that average, some of the particles will have a much lower energy (they are moving or vibrating or whatever more slowly); some of the particles will have a much greater energy; most particles will have energies somewhere in between. Every time two particles collide, their energy due to movement will change. One particle will probably gain energy, and the other will lose it. The total amount of energy as a result of the collision will stay the same, but it has been redistributed between the particles. That is happening throughout the whole substance. But if you could work out the average energy of the particles due to their movement, it will always be the same as long as the temperature stays the same. What has this got to do with evaporation? Suppose one of the particles in the surface of the liquid was suddenly given some more energy because of a collision. Such a particle is coloured red in the first diagram below.
That particle may well now have enough energy for it to break the attractions to its neighbours, and escape from the surface. And the same thing can happen to other particles in the surface as well - as in the second and third diagrams. The liquid is gradually evaporating. | |
|
A couple of points: I have left the particles painted red after they have escaped from the surface, not because they have chemically changed in any way, but because they are still relatively energetic. In reality, they will probably very soon collide with air particles over the liquid, and may well transfer that extra energy to one of those. If you are really wide awake, you might work out that if you are losing especially energetic particles from the surface, the average energy of those remaining will fall. Does that mean that the temperature of the liquid will fall? Yes! If you put a drop or two of a volatile liquid (one that turns to a gas very easily - such as alcohol or ether) on your skin, your skin will feel cold. The liquid is cooling because it is losing its most energetic particles, and is taking heat energy from your skin to warm it up again. Any evaporating liquid will take heat energy from its surroundings as it cools. Heat energy flows from a warmer to a cooler place. That constant flow of heat energy into an evaporating liquid supplies the energy for still more particles to break away from the surface. | |
|
Does water in a sealed bottle evaporate? If you had some water in a bottle with a really good sealed cap, you could come back decades later and it wouldn't look any different. So why isn't it evaporating? Actually, it is! The difference here is that the particles of water which break away from the surface are bouncing around in the space above the liquid - they can't escape, and sooner or later will hit the surface of the water again. They could, of course, just bounce off again if they happened at that moment to be particularly energetic (because of recent collisions they have had in the gas), but it is perhaps more likely that they will be trapped again by attractions with the particles in the surface. After a very short time, you get an equilibrium situation where the number of particles leaving the surface is exactly balanced by those sticking back on the surface. Once that equilibrium is set up, nothing more happens on the grand scale - although there is a constant interchange of particles between the surface and the gas on a much smaller scale. We call this a dynamic equilibrium. The word dynamic stresses the fact that there is constant interchange of water particles between the liquid and the gas. It isn't particularly important for now, but you will meet this idea again later on during your course in a more chemical context. Changes of state between solid and gas - sublimation The official definition of sublimation from IUPAC (the International Union of Pure and Applied Chemistry) is the direct transition of a solid to a vapour without passing through a liquid phase Solid carbon dioxide - dry ice Dry ice (solid carbon dioxide) has a temperature of below -78.5°C. It is a white solid, and very cold. Why dry ice? Because it never forms a liquid. If dry ice warms up, at -78.5°C the solid carbon dioxide starts to convert to carbon dioxide gas. There is no such thing as liquid carbon dioxide at ordinary pressures. This conversion from solid straight to gas without first forming a liquid is called sublimation. Iodine In a simple lab situation you are probably more likely to come across iodine as an example of sublimation, but there is a real problem in discussing iodine sublimation. Let's look at a typical example from YouTube. At the beginning of the video clip, you can clearly see that some of the iodine has gone from solid into a gas, and later on you can clearly see that iodine has crystallised straight from the gas into a solid on the cooled watch glass without in either case going through liquid iodine. The problem comes in the middle when the gas is so densely purple in the beaker that you can't actually see what is happening to the iodine. In fact, iodine melts at 113.5°C and then boils at 184.4°C. So when it is being heated, it isn't necessarily going from solid straight to gas. Another piece of video also from YouTube shows what happens if you control the temperature carefully at about 140°C. So does iodine sublime? Well, it certainly goes straight from solid to gas at temperatures below its melting point. It certainly goes from gas to solid when the gas is cooled. But it can also go via the normal route of solid melting to liquid and then ending up as gas. So it does sublime, but it doesn't always sublime. | |
|
Note: It is important that you find out what your examiners want you to say about this - at least one UK GCSE syllabus specifically mentions the sublimation of iodine. If you are just asked for an example of sublimation, give dry ice - there is nothing awkward about its behaviour. If you are doing a UK-based syllabus, you can find links to the Exam Boards' websites where you can download a copy of your syllabus and other useful stuff on the about this part of Chemguide page. | |
© Jim Clark 2018 |
|