|
Chemguide: Core Chemistry 14 - 16 An introduction to fractional distillation in the oil industry This page talks about how the complex mixture of hydrocarbons in crude oil (petroleum) can be separated into simpler mixtures using fractional distillation. What is crude oil (also known as petroleum)? The photo (taken from Wikipedia) shows a sample of crude oil.
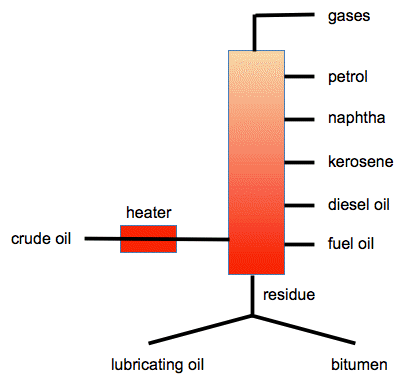
Crude oil is a fossil fuel. It was formed over millions of years as living things in ancient seas died, fell to the bottom and got covered with other sediments. Over time, as the sediments built up, the once-living matter was subjected to high pressures and temperatures, and was converted into what we now know as crude oil or petroleum. It is a complicated mixture mainly of hydrocarbons - compounds of carbon and hydrogen only. These range from simple ones like methane, CH4, through moderately sized ones like octane, C8H18, up to very big ones which contain up to about 70 carbon atoms (found in bitumen). Fractional distillation Fractional distillation is the process used to split the complex mixture of hydrocarbons into simpler mixtures of hydrocarbons (called fractions) depending on their boiling points. Words of caution! It is essential that you find out exactly what your examiners want. Most, but not all, of the UK syllabuses give a list of fractions with their uses, and that list differs between syllabuses. It is also important to look at past exam papers and mark schemes so that you know what depth you need to go into. In my experience most questions on this topic are pretty trivial. You will find links to the websites of most of the UK Exam Boards on this page. A summary diagram The crude oil is heated to about 400°C when most of it turns to vapour. The vapour is passed into a tall column which gradually gets cooler towards the top. The vapours rise until they get to a point where the temperature is cool enough for them to condense. Mixtures of hydrocarbons with similar boiling points are tapped off at intervals up the column. This diagram is based on most other diagrams you will find.
The residue is the part of the original crude oil which wasn't turned into a vapour by the heater. The top of the tower is likely to be at a temperature of about 25 - 40°C, and the bottom at about 350 - 400°C. The safest thing is to use any numbers given in a textbook endorsed by your Exam Board. Adapt the diagram so that it only includes fractions mentioned by your syllabus. For example, not all syllabuses mention naphtha - leave it out if it isn't on yours. Only one syllabus mentions lubricating oil. In all other cases, you could just show bitumen being produced from the bottom of the tower. Some trends in properties of the fractions Molecular size The molecular size increases as you go from gases to the other fractions down though the column. The gases have small molecules with up to 4 carbon atoms. Bitumen has very large molecules containing up to about 70 carbon stoms. Most of the other properties below depend on the sizes of the molecules. Boiling points Obviously, we have just separated the fractions using their different boiling points. The ones collected further down the column condensed to liquids at much higher temperatures than those collected further up. The boiling points of the fractions increase as you go down the column. The reason for this lies in the size of the molecules influencing the intermolecular forces. Intermolecular forces are the forces which attract one molecule to another one. The intermolecular forces in hydrocarbons are van der Waals dispersion forces which increase in size as the molecules get longer with more electrons. Volatility The fractions containing small molecules are more volatile than those containing bigger ones. Volatility is just a way of talking about how easily a liquid turns to a vapour. It is pretty obvious if you think about it. Petrol is very volatile - it evaporates quickly if it is left exposed to air. Bitumen (used in tar on roads) is obviously not volatile at all. This is again due to the increasing strengths of the intermolecular forces as the molecules get bigger. Flammability This is important. Flammability is a measure of how easy it is for the hydrocarbon to catch fire. It is obvious that petrol catches fire very easily, whereas if you drop a lighted match on a bit of road tar, nothing happens. So the ease of combustion falls as you go from gases to petrol to the rest. This is related to the volatility of the hydrocarbon. Petrol is dangerously flammable, because in the air over the top of a petrol spill, for example, you have a very reactive mixture of petrol vapour and air. Colour As you go down the column, the fractions become darker in colour - there's not much more you can say about that! Viscosity Viscosity is a measure of how easily a liquid flows. Something which has a low viscosity is very runny; something which has a high viscosity is much more sticky. So a viscous liquid is sticky; the simplest opposite of viscous is runny. Again, petrol is obviously very runny whereas bitumen definitely isn't. The liquids get more viscous as you go to fractions lower in the column. This is again because ot the increase in intermolecular forces as the molecules get bigger. If the molecules have more attractions between each other, they aren't going to flow very easily. Some uses of the fractions Important! Don't waste time learning uses for fractions which aren't on your syllabus. Refinery gases Main source of propane and butane used as liquefied petroleum gas (LPG) for cooking and heating. Petrol (gasoline) fraction As a fuel for cars. Naphtha fraction An important source of organic compounds for the chemical industry. Kerosine fraction Kerosine is also known as paraffin. Mainly used as a jet fuel. Diesel oil fraction Also known as gas oil. As a fuel for cars, lorries, buses and trains powered by diesel engines. Fuel oil fraction There are various grades of this. Heavy fuel oil is used in ships' boilers. Lubricating oil fraction Further processing leads to lubricants, waxes and polishes. Bitumen Used in road making.
© Jim Clark 2021 |