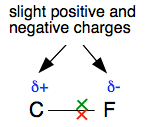|
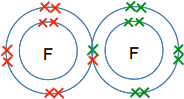
Chemguide: Core Chemistry 14 - 16 Polar covalent bonds This page introduces the idea of electronegativity and how electronegativity differences between atoms leads to polar covalent bonds - bonds which aren't electrically neutral, but have a slightly positive and a slightly negative end. I am assuming that you have read the page introducing covalent bonding and have access to a Periodic Table such as the one you can download from this site. The download button is at the beginning of the second paragraph under the table. An introduction to electronegativity Suppose you had two fluorine atoms joined together by a covalent bond - a pair of shared electrons.
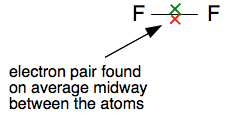
Just showing the bonding pair on a line drawing , , ,
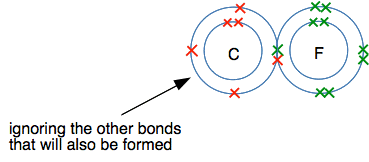
The electrons are moving, but on average they will be found half-way between the two nuclei. There is nothing to attract them permanently to one end or the other - the two ends are identical. Now replace one of the fluorines by a carbon atom to make a carbon-fluorine covalent bond. We aren't interested in what else is attached to the carbon as well - so I am just leaving them out.
A line diagram shows that something important has happened:
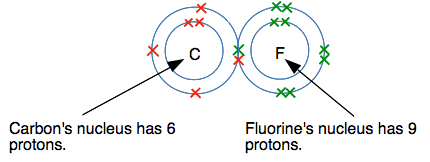
The bonding pair has now been drawn closer to the fluorine, and is no longer half-way between the two nuclei. Defining electronegativity Electronegativity is a measure of an atom's ability to attract a bonding pair of electrons. Fluorine has a higher electronegativity than carbon, and so attracts the bonding pair towards itself. Why has fluorine got a higher electronegativity than carbon? This is really a question for a higher level of chemistry, but it isn't very difficult to understand and will be helpful for the next topic - so it is worth a quick look. Look at the dots-and-crosses diagram again, but this time including the numbers of protons in the two nuclei.
In fact, the bonding pair of electrons doesn't feel the full pull of these protons because it is shielded (or screened) from them to some extent by the two electrons in the inner level. Taking that into account, there is, very approximately, a net pull on the bonding pair of 4+ from the carbon side (6 protons less the effect of the 2 inner electrons), but 7+ from the fluorine side. Obviously the bonding pair will be pulled closer to the fluorine. The negative electrons are more strongly attracted to 7 positive charges than to 4. Polar covalent bonds Look again at the bonding pair of electrons in the C-F bond.
The carbon's electron has moved away to some extent from where it was in the original atom towards the fluorine. But there has been no change in the position of the nucleus and the protons. That means that the positive charge due to the protons isn't quite as balanced as it was by the negative charge due to the electrons around it. The net effect is that the carbon end of the bond is left slightly positive. On the other hand, the fluorine has gained a bit of extra negative charge due to carbon's electron getting closer to it. We show these slight positive and negative charges in a diagram by using the symbol δ (say "delta"). So a slight positive charge is δ+ ("delta plus") and a slight negative charge is δ- ("delta minus").
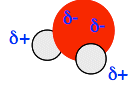
This is a polar bond because it has an electrical dipole - a separation of charge between one end and the other. Polar covalent bonds are very common, because the electronegativities of the two atoms at either end of the bond are very unlikely to be the same unless both atoms are the same. With two different atoms involved, one is almost certain to attract a bonding pair of electrons more than the other one does. But at this level, there is only one example that you are likely to come across - and that's water. Oxygen is more electronegative than hydrogen, and so attracts the bonding pair of electrons towards itself. That happens in both O-H bonds. The effect is that the oxygen atom has some extra negative charge on it, and each hydrogen has a slight positive charge.
The polarity of the water molecule is really important in chemistry, and the place you will meet it next is when we look at why things are soluble (or not) in water.
© Jim Clark 2019 |