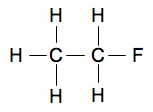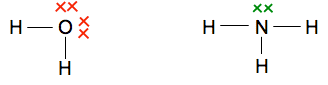|
Chemguide: Core Chemistry 14 - 16 Introducing covalent bonding This page introduces the way atoms can bond together by sharing electrons in single covalent bonds, and the various ways you can show covalent bonds on paper. I am assuming that you can work out electronic structures of simple atoms and have access to a Periodic Table such as the one you can download from this site. The download button is at the beginning of the second paragraph under the table. Why do atoms form bonds? Suppose you had two hydrogen atoms joined strongly together by some means. If you wanted to break the bond to give two separate hydrogen atoms again, it would cost you quite a lot of energy. If you allowed the hydrogen atoms to join up again, because you are simply reversing what you just did, you will get that same amount of energy out again. So breaking bonds costs energy, and making bonds releases energy. That is illustrated in the diagram:
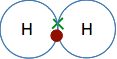
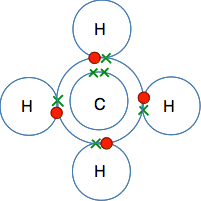
By losing that amount of energy when they combine, the hydrogen molecule (with the two atoms joined together) is energetically more stable than the separated atoms. Atoms combine together make a molecule because energy is released and the molecule is energetically more stable than its separate atoms. A simple covalent bond in the hydrogen molecule, H2
The electrons from the two hydrogen atoms come together to make a pair, and it is this pair of shared electrons between the two nuclei that forms the covalent bond. So what is actually holding the atoms together? The nucleus of the left-hand hydrogen atom is strongly attracted to the electron pair; the nucleus of the right-hand hydrogen atom is also attracted to the same electron pair. It is these two attractions to the same pair of electrons that acts as the "glue" holding the atoms together. | |
|
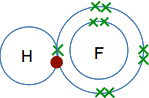
Note: Diagrams like this are often described as "dots-and-crosses" diagrams. It is usual to show the electrons from the two atoms differently so that you can tell which came from which atom. You can use dots for one atom and crosses for the other (as here). You can use differently coloured crosses for the two atoms; or you can use differently coloured dots for the two atoms. It doesn't matter as long as it is clear. But be careful! Some examiners don't like you to use colour in your answers. To be on the safe side, just use dots for one atom, and crosses for the other in pencil or whatever blue or black pen you use for writing everything else. | |
|
Some other simple examples Hydrogen fluoride, HF Again, an electron pair is shared - there was a single electron on the original hydrogen atom and also one on the fluorine atom.
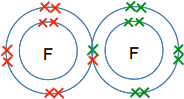
Fluorine, F2 Just like hydrogen, fluorine consists of molecules made up of two atoms rather than going around as single atoms. Why? Because the formation of the bond between the atoms releases energy, and the molecule is energetically more stable than individual atoms.
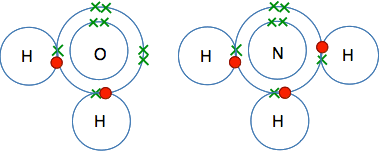
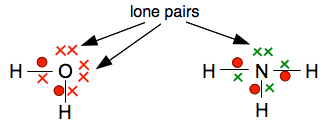
Why do they go around in pairs rather than some other number? Because each atom only has one unpaired electron which it can share with another one. The molecule formed has no unpaired electrons for the process to go any further. Water, H2O, and ammonia, NH3 In both oxygen and nitrogen, there is more than one unpaired electron. That means that each of these can form more than one covalent bond. They combine with enough hydrogen atoms so that all the unpaired electrons are converted into covalent bonds. That maximises the amount of energy released and so makes the molecules as energetically stable as possible.
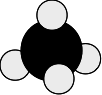
Methane, CH4 This doesn't add anything much to the discussion, but it is a frequently discussed molecule, and I include it for completeness. Carbon has 4 unpaired electrons, and so can form 4 covalent bonds with hydrogen atoms.
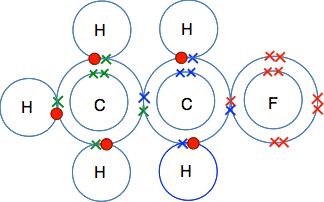
A more complicated molecule, C2H5F I have chosen this because we have already got all the dots-and-crosses diagrams for C, H and F further up the page. If you have 2 carbon atoms, 5 hydrogens and a fluorine atom, there is only one way you can fit them together so that all the electrons are paired, and all the atoms are held together by shared pairs of electrons.
Look carefully at this, and make sure you can see where all the electrons are coming from, and how they produce a whole lot of covalent bonds. Now do one yourself: C2H6O On a piece of paper, draw the electronic structures of 2 carbon atoms, 6 hydrogens and an oxygen atom, and find a way to bond them together so that there are shared electrons between each atom and its neighbour(s), and there are no unpaired electrons left over. There are actually two different ways of joining these up to make two different molecules. If you find one, good! If you find both, excellent! When you have a solution, click here to check your answer. Quicker ways of showing covalent bonds in molecules Just showing the outer electrons in dots-and-crosses diagrams In the examples you will meet at this introductory level, it is only the electrons in the outer energy level which are involved in the bonding. That means that you could leave out the inner levels - but, in an exam, only if you are given permission in the question. So that instead of drawing all three levels in chlorine (2,8,7), you could only draw the outer level with the 7 electrons in it. Showing the bonds by drawing lines between the atoms In chemistry the convention is that any line that you draw between two atoms represents a covalent bond - a pair of shared electrons. So, for example, the molecule of C2H5F that we drew in full above . . .
. . . could be simply shown as:
This will be by far the most common way of showing covalent bonds that you will meet during the course. Showing the bonds by drawing lines but adding some electrons as well For example, water, H2O, and ammonia, NH3, that we drew above in a dots-and-crosses diagram . . .
could be shown:
These show the covalent bonds with lines, but also show all the outer electrons as well. The pairs of electrons in the outer levels which aren't used for bonding are known as lone pairs, and are important in the chemistry of both water and ammonia. This isn't a common way of drawing these diagrams. It is more likely, where you need to emphasise the lone pairs, to draw the diagrams just with the lines and the lone pairs. Putting the electrons on the lines is really just saying the same thing twice - both show a covalent bond. So the last diagram could be simplified to:
Do dots-and-crosses diagrams tell you about the shapes of the molecules? The short answer is - no, not directly. Earlier on this page, you met the dots-and-crosses diagram for methane:
Taken literally, that would suggest that methane is a flat molecule, but it isn't. What happens is that all the electron pairs in the outer level of the carbon arrange themselves so that they can get as far apart from each other as possible. So the actual shape of the molecule is:
And ammonia, NH3, and water, H2O, molecules look like this:
If you look carefully, they are all based on the same shape as the methane. The missing hydrogens are replaced by invisible lone pairs of electrons. You don't need to worry about any of this at this introductory level. I include it just for interest.
© Jim Clark 2019 |
|