|
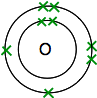
Chemguide: Core Chemistry 14 - 16 Double and triple covalent bonds This page introduces the possibility of multiple (double or triple) covalent bonds between atoms using oxygen, nitrogen, carbon dioxide and ethene as examples. These are chosen because they are all commonly specified on UK syllabuses for 14-16 year olds. I am assuming that you have read the page introducing covalent bonding and have access to a Periodic Table such as the one you can download from this site. The download button is at the beginning of the second paragraph under the table. Double bonding in oxygen If you look up oxygen on the Periodic Table, you will find that it has an atomic number of 8 - and so the neutral atom has 8 protons and 8 electrons. These will be arranged 2,6. So you have two atoms like this:
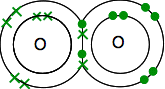
Each has two unpaired electrons. To get the maximum energetic stability, all of these will need to be paired up. So the oxygens share two pairs of electrons:
This is much easier to see using lines to represent pairs of shared electrons:
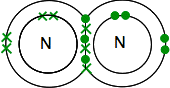
Triple bonding in nitrogen If you find nitrogen in the Periodic Table, you will be able to work out that its electronic structure is 2,5. Comparing it with oxygen, it therefore has one pair of electrons and three unpaired electrons in its outer level. To make it as energetically stable as possible, it will need to pair all of these up in covalent bonds. Two nitrogen atoms will therefore combine in this way:
Again, this is much easier to see using lines to represent pairs of shared electrons:
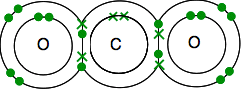
Double bonding in carbon dioxide Carbon's electronic structure is 2,4 with 4 unpaired electrons in the outer level. Oxygen's is 2,6 with 2 pairs of electrons and 2 unpaired electrons in the outer level. You need to end up with all the electrons paired - and there is only one way of doing it.
As before, the bonding is easier to see using lines for bonding pairs of electrons.
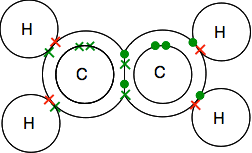
If there are two bonding pairs of electrons between two atoms, it is called, not surprisingly, a double bond. So each oxygen is joined to the carbon atom in the middle by a double bond. Double bonding in ethene, C2H4 Ethene and other similar compounds with double bonds between carbon atoms are important substances in chemistry. You will meet them in more detail later in the course. Look carefully at the dots-and-crosses diagram and be sure you understand how it all fits together. It has to be this way so that all the single electrons in both the carbons and the hydrogens are paired up in bonding pairs.
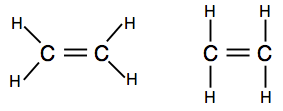
If you come across diagrams showing the bonding pairs as lines, you may find one of two versions.
The first one shows the actual shape of the molecule better, but the second is also perfectly acceptable at this level.
© Jim Clark 2019 |