|
Chemguide: Core Chemistry 14 - 16 The Bronted-Lowry theory of acids and bases. There are three different theories of what acids and bases are, but the most useful one at this level is the Bronsted-Lowry theory. If you have worked through the various reactions of acids and bases in this section, then you will have met almost all of the reactions you need to know about. This page just tidies things up. What is the Bronsted-Lowry theory?
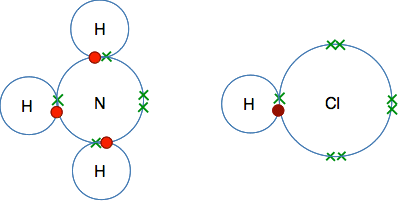
The reaction between ammonia gas and hydrogen chloride gas Let's explore dots-and-crosses diagrams of the two molecules. To avoid clutter, the diagrams only show the outer electrons. Here are ammonia and hydrogen chloride molecules before reaction:
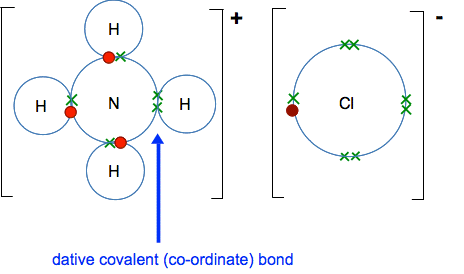
And this is what has happened after reaction:
A hydrogen ion has transferred from the chlorine to the lone pair on the nitrogen atom. Notice that the hydrogen has left its electron behind with the chlorine. That extra electron in the chlorine's outer level means that it is now a chloride ion. In the HCl, the charge on that electron was balanced by the proton in the hydrogen's nucleus. The new bond between the nitrogen and the incoming hydrogen is formed with both electrons coming from the same atom. That is a co-ordinate (or dative covalent) bond. Because the hydrogen came as a nucleus without its electron, the ammonium ion formed has one more positive than negative charge, and so carries an overall positive charge. | |
|
Note: If you aren't happy about co-ordinate (dative covalent) bonds follow this link. | |
|
So . . .
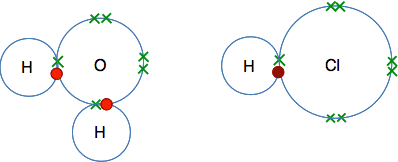
Acids in solution If you have read the other pages in this section, you will know that acids in solution contain hydrogen ions, which we have described in equations as H+(aq). At this level, that is probably all you need to know, but I want to explore this a bit more. The important thing to notice is that there is no such thing as a simple hydrogen ion in chemical reactions. A hydrogen ion is just a proton with no electrons associated with it. It has a full positive charge concentrated into a very small space and is immensely reactive. In chemistry, the hydrogen ion is always attached to something else. In water, the hydrogen ion is attached to a water molecule in exactly the same way as the donated hydrogen is attached to the ammonia molecule above. So let's look at hydrogen chloride gas dissolving in water to make hydrochloric acid. This is what we are starting with:
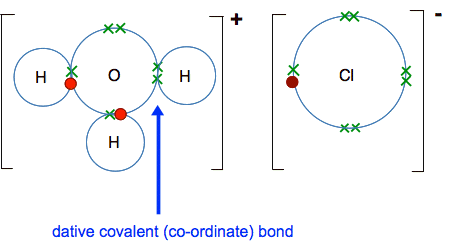
And after the same donation of a hydrogen ion as before, you get:
In equation form: H2O(l) + HCl(g) The H3O+ ion is called a hydroxonium ion, and this is how hydrogen ions occur in solution. Whenever you write H+(aq) this is what you are actually talking about. Notice that the equation that we have just written is also an example of an acid-base reaction in the Bronsted-Lowry theory. The HCl has donated a hydrogen ion to the water and so is acting as an acid. The water has accepted the hydrogen ion and so is acting as a base. This would be true of any acid in water - the acid will donate a hydrogen ion to a water molecule and form a hydroxonium ion. Reactions of acids in solution with metal oxides, hydroxides and carbonates On another page we talked about most metal oxides and hydroxides as being bases and used these ionic equations:
Obviously the oxide ions or the hydroxide ions are acting as bases because they are accepting hydrogen ions and combining with them to make a new product, water. When we were talking about carbonates, we used this ionic equation:
The carbonate ions are acting as bases because they are accepting hydrogen ions and then breaking up to form new products. Going beyond what you need for this level Because we have looked at the formation of hydroxonium ions when acids react with water, let's just finish off with a slightly more accurate view of the last two equations. What the equations show as H+(aq) is, of course really a hydroxonium ion, H3O+(aq). It is that ion which is actually acting as the acid donating hydrogen ions to the oxide or hydroxide or carbonate ions. Not all reactions of acids are acid-base reactions! The acid-base reactions we have looked at are:
But we haven't made any mention of acid + metal. That's because this isn't an acid-base reaction. If, for example, magnesium reacts with an acid the ionic equation is
The magnesium atoms aren't accepting hydrogen ions and combining with them in some way. What is happening is simply a transfer of electrons from the magnesium to the hydrogen ions. The magnesium loses two electrons to make a magnesium ion. The hydrogen ions gain those electrons to make hydrogen atoms which combine to make hydrogen molecules. This is a redox reaction. If you aren't sure about this, you might want to refer back to this page. Two other theories of acids and bases I am just mentioning these because you might come across one of them at this level. You would only meet the second one in a more advanced course. The Arrhenius theory This is a very old definition which has been overtaken by the Bronsted-Lowry theory On the Arrhenius theory:
This doesn't go against the Bronsted-Lowry theory - it is just much more limited. The Lewis theory This extends the Bronsted-Lowry to cases which you wouldn't normally think of as acid-base reactions. I'm not going to talk about this at all. If you are interested google lewis acids and bases.
Again, the Lewis theory doesn't stop you using the Bronsted-Lowry theory - it just makes it much more general (and I mean much more general!).
© Jim Clark 2020 |
|