|
Chemguide: Core Chemistry 14 - 16 Reactions between metals and dilute acids This page expands on work already covered in the Reactivity Series section and assumes you are already familiar with the reactions between metals and acids from that. If you haven't very recently read the page the reactions between metals and acids in that section, go and read it before you continue with this page. Use the back button on your browser to return here when you are happy with that. What you should be comfortable with before you go on
| |
|
Note: When you are trying to remember descriptions of reactions, find the general pattern, and then just learn those which are different.. For example, remember that reactions get faster the further up the Reactivity Series you go, but that aluminium is very slow to start with and then speeds up. Most metals react to give hydrogen and a colourless solution, but iron gives a very pale green solution. If you can recognise patterns like this, it makes life much easier than trying to learn each one separately. | |
|
A summary equation For most acids: metal + acid Remember that a salt is formed when the hydrogen in an acid is replaced by a metal or an ammonium group. | |
|
Note: Although you are unlikely to meet the problem at this level, you should know that nitric acid behaves differently, for reasons we wil touch on further down this page. | |
|
Taking a closer look at two of the metals commonly reacted with acids Magnesium Magnesium reacts with dilute sulfuric acid to give a colourless gas, hydrogen, and a colourless solution of magnesium sulfate.
The reaction with dilute hydrochloric acid looks exactly the same, but this time magnesium chloride is produced.
Why are they so similar when you are producing two different things? Acids in solution contain hydrogen ions, H+(aq). In addition, dilute sulfuric acid contains sulfate ions, SO42-(aq). Hydrochloric acid also contains hydrogen ions, this time accompanied by chloride ions, Cl-(aq). In the salts formed (magnesium sulfate and magnesium chloride), the negative ions are completely unchanged - they were swimming around in the solution before, and are still swimming around after the reaction has happened. But the original magnesium atoms have, in both cases, turned into magnesium ions, Mg2+(aq). You can write the ionic equation for the reaction showing what is really happening, leaving out the unchanged negative ions - known as spectator ions.
And that is the same for both sulfuric acid and hydrochloric acid. The reactions look the same, because they both involve exactly the same bit of chemistry. | |
|
Important: If you aren't familiar with the terms oxidation, reduction, and redox reactions, you need to read the page An introduction to redox reactions before you go on. Return here using the back button on your browser when you have finished. | |
|
This is a redox reaction. A redox reaction is one in which reduction and oxidation are happening. Electrons are being transferred in this case. Remember OIL RIG - oxidation is loss of electrons; reduction is gain of electrons. Magnesium atoms are losing electrons to make magnesium ions. This is oxidation. The hydrogen ions are gaining electrons to make hydrogen molecules and so are being reduced. Zinc The situation is exactly the same in the reactions between zinc and the two acids.
The sulfate ions or chloride ions are again spectator ions, and the ionic equation for the two reactions is just like the magnesium reactions.
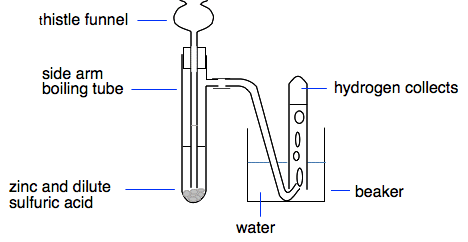
Zinc atoms are losing electrons to make zinc ions. This is oxidation. The hydrogen ions are gaining electrons to make hydrogen molecules and so are being reduced. The sulfate or chloride ions are entirely unaffected. Using the zinc and dilute sulfuric acid to make hydrogen in the lab This is a traditional way of producing small amounts of hydrogen in the lab.
Zinc is a bit slow to react with dilute acids in the cold, and a small amount of copper(II) sulfate solution is added to speed up the reaction. It is often said that the copper(II) sulfate is added as a catalyst, but that isn't strictly true. A catalyst is chemically unchanged at the end of a reaction - but the copper(II) sulfate is changed. There is a reactivity series reaction between the copper(II) sulfate solution and the zinc, producing a little bit of copper. It is the presence of copper which actually catalyses the reaction. A brief comment about the problem with nitric acid This is for interest only - you are very unlikely to be asked about this in an exam at this level. You have seen that in the reactions between metals and dilute acids, the metal is oxidised to metal ions by the hydrogen ions in the acid, and the hydrogen ions are reduced to hydrogen gas. The problem with nitric acid, HNO3, is that a nitrate ion, NO3-, is a much stronger oxidising agent than a hydrogen ion. So the nitrate ions oxidise metal atoms to give metal ions, but don't give hydrogen as well. Instead, when the nitrate ions are reduced, they give oxides of nitrogen - NO or NO2. Which you get depends on the concentration of the acid. You can get hydrogen from very dilute nitric acid with a reactive metal like magnesium, but even in that case you will get some nitrogen oxides formed as well. Remember: This is for interest only. Unless your syllabus specifically mentions reactions between metals and nitric acid, you don't need to know this. If it does mention them, then you will have to find another source of the detail you might need. If you are doing a UK-based syllabus, you can find links to the Exam Boards' websites where you can download a copy of your syllabus and other useful stuff on the about this part of Chemguide page. Strong and weak acids All of the acids we have looked at so far on this page are strong acids, and we need to define what that means. If you talk about strong tea, for example, you mean a cup of very dark tea with a lot of tea dissolved in it. In chemistry, that would be described as concentrated tea.So a solution of an acid containing a high or very high proportion of acid would be described as a concentrated acid. A strong acid means something much more sophisticated. When we talked about sulfuric acid or hydrochloric acid or nitric acid above, we talked about the acid being ionised to give hydrogen ions in solution together with a negative ion (sulfate, chloride or nitrate).
You can, of course, have a concentrated solution of a strong acid or a dilute solution of a strong acid. The word "strong" is independent of the concentration. By contrast, a weak acid is only partially ionised in solution. A common example of a weak acid is ethanoic acid (acetic acid), CH3COOH. This ionises to a very small extent in solution to give ethanoate (acetate) ions and hydrogen ions.
The new arrows show that the reaction is reversible. The molecules are forming ions, and the ions are coming together again to re-form the molecules. The net effect is that at any time most of the acid is present as molecules, and only a small proportion as ions.
Again, you can have a concentrated solution of ethanoic acid or a dilute solution of ethanoic acid. Concentrated and dilute are measures of how many molecules of acid there are in the solution. Strong and weak are measures of what proportion of those molecules are ionised. How do the reactions of weak acids compare with those of strong acids? Reactions of acids in solution are governed by the hydrogen ions present. If there are fewer hydrogen ions, the reactions will be slower. So if you add magnesium ribbon to ethanoic acid, it will be slower than the reaction between magnesium and sulfuric acid, for example. And that's it! You will still get a salt and hydrogen - just more slowly. You will meet some chemistry of ethanoic acid when you do organic chemistry, and I will leave the details until then. | |
|
Note: You might perhaps think that if there are only a small number of hydrogen ions in solution, the reaction would soon stop. You would be wrong! If the hydrogen ions are removed by reacting with something, they can't react with the ethanoate ions again to re-form the ethanoic acid. But there is nothing to stop the ethanoic acid molecules from ionising. The net effect is that every time a hydrogen ion reacts, it is replaced by another one. But the concentration will still be low, and so the reaction is still slow. | |
© Jim Clark 2020 |
|