|
Chemguide: Core Chemistry 14 - 16 The physical properties of molecular substances This page describes how the physical properties of substances having molecular structures varies with the type and strength of the intermolecular attractions. I am assuming that you already know about intermolecular forces. The current page builds on that page, and so it is essential that you have read it recently. What are molecules? Molecules are made of fixed numbers of atoms joined together by covalent bonds, and can range from the very small (even down to single atoms, as in the noble gases) to the very large (as in polymers, proteins or even DNA). The covalent bonds holding the molecules together are very strong, but these are largely irrelevant to the physical properties of the substance. Physical properties are governed by the intermolecular forces - forces attracting one molecule to its neighbours. These could be dipole-dipole attractions between polar molecules, or van der Waals dispersion forces (due to temporary fluctuating dipoles), or hydrogen bonds - or some combination of them. | |||||||||||||
|
Important! If you don't understand that last sentence, there is no point in you continuing with this page. Read the page about intermolecular forces first. | |||||||||||||
|
Melting and boiling points Molecular substances tend to be gases, liquids or low melting point solids, because the intermolecular forces of attraction are comparatively weak. You don't have to break any covalent bonds in order to melt or boil a molecular substance. | |||||||||||||
|
Note: This is really important! You can make yourself look extremely stupid if you imply in an exam that boiling water, for example, splits it into hydrogen and oxygen by breaking covalent bonds. Exactly the same water molecules are present in ice, water and steam. | |||||||||||||
|
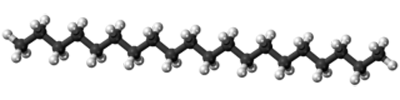
The size of the melting or boiling point will depend on the strength of the intermolecular forces. The presence of hydrogen bonding will lift the melting and boiling points. The larger the molecule the greater the attractions due to van der Waals dispersion forces (temporary fluctuating dipoles) - and those will also need more energy to break. Three molecules which only have dispersion forces For example, here are three hydrocarbons (diagrams all taken from Wikipedia):
Here are their melting and boiling points:
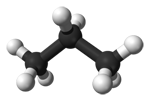
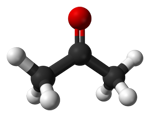
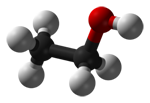
You can see that as the molecules get bigger, so do their melting and boiling points. These molecules aren't polar, and they don't have hydrogen atoms attached directly to an oxygen, nitrogen or fluorine atom, and so they don't have hydrogen bonding. The intermolecular attractions are due only to dispersion forces, and these increase as the number of electrons and the length of the molecules increase. Three molecules which have a variety of intermolecular forces Here are three more molecules of much the same size this time, but with different sorts of intermolecular forces involved in each case. (Diagrams again from Wikipedia.)
The only intermolecular forces in propane are dispersion forces (due to temporary fluctuating dipoles). Because the molecule is quite small, these are fairly weak. It boils at -42°C. Propanone has a dipole because ot the polarity of the carbon-oxygen bond, and so it has permanent dipole-dipole attractions as well as the temporary ones. It boils at 56°C. Ethanol has hydrogen bonding because ot the hydrogen attached directly ot the oxygen. It also has a permanent dipole as well as the ever-present dispersion forces. It boils at 78°C. Summary There is a wide variation in melting points and boiling points of molecular substances, depending on how big they are, and exactly what sorts of intermolecular attractions are present. The important thing to remember at this level is that molecular substances tend to be gases, liquids or low melting point solids. The size of the melting or boiling point will depend on the strength of the intermolecular forces. The presence of hydrogen bonding will lift the melting and boiling points. The larger the molecule the more van der Waals attractions are possible - and those will also need more energy to break. Solubility in water Most molecular substances are insoluble (or only very sparingly soluble) in water. Those which do dissolve often react with the water, or else are capable of forming hydrogen bonds with the water. Why doesn't a gas like methane, CH4, dissolve in water? The methane itself isn't the problem. Methane is a gas, and so its molecules are already separate - the water doesn't need to pull them apart from one another. The problem is the hydrogen bonds between the water molecules.
There are quite strong hydrogen bonds between the fairly positive hydrogens and the lone pairs on the fairly negative oxygens. If methane were to dissolve, it would have to force its way between water molecules and so break hydrogen bonds. That costs a reasonable amount of energy. The only attractions possible between methane and water molecules are the much weaker van der Waals dispersion forces - and not much energy is released when these are set up. It simply isn't energetically profitable for the methane and water to mix. So non-polar molecules don't tend to dissolve in water. Why is ammonia gas, NH3, very soluble in water? Ammonia has the ability to form hydrogen bonds. When the hydrogen bonds between water molecules are broken, they can be replaced by equivalent bonds between water and ammonia molecules. Some of the ammonia also reacts with the water to produce ammonium ions and hydroxide ions, but at any one time only about 1% of the ammonia has actually reacted to form ammonium ions. The solubility of ammonia is mainly due to the hydrogen bonding and not the reaction. Other common substances which are freely soluble in water because they can hydrogen bond with water molecules include ethanol (alcohol) and sucrose (sugar). Solubility in organic solvents Molecular substances are often soluble in organic solvents - which are themselves molecular. Both the solute (the substance which is dissolving) and the solvent are likely to have molecules attracted to each other by van der Waals dispersion forces. Although these attractions will be disrupted when they mix, they are replaced by similar ones between the two different sorts of molecules. Electrical conductivity Molecular substances won't conduct electricity. Even in cases where electrons may be delocalised within a particular molecule, there isn't sufficient contact between the molecules to allow the electrons to move through the whole solid or liquid.
© Jim Clark 2019 |
|||||||||||||