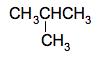|
Chemguide: Core Chemistry 14 - 16 Alkanes This page introduces alkanes and the important term "homologous series". It looks at the combustion of alkanes and their reactions with chlorine and bromine. I am assuming that you have already read the pages about organic formulae and names and about isomerism. Homologous series There are huge numbers of organic compounds, but they fall into families known as homologous series. All the members of a homologous series have the same type of bonding. The homologous series of the alkanes only has carbon-hydrogen bonds and carbon-carbon single bonds apart from methane, CH4, the first member of the series which only has carbon-hydrogen bonds. All members of a homologous series . . .
For example: The alkanes have a very limited chemistry. The only simple things they do are burn or react with chlorine or bromine. All alkanes have the same reactions because they all have the same sort of bonds. Reactions happen when you break bonds. The boiling points of the alkanes increase in a regular way. The first four alkanes (up to butane, C4H10) are gases at room temperature. Then you get liquids as the boiling points increase, and eventually solids when they get really big. You really need to remember that the first four are gases. A general formula lets you work out the formula of any member of a homologous series if you know the number of carbon atoms. For alkanes, the general formula is CnH2n+2 So, for example, methane with one carbon atom has (2 x 1) + 2 carbon atoms so the formula is CH4. Octane with 8 carbon atoms has (2 x 8) + 2 carbon atoms so the formula is C8H18. Notice that as you go from one alkane to the next, the formula increases by CH2. The alkanes The alkanes are saturated hydrocarbons. A saturated hydrocarbon is one which has the maximum possible number of hydrogens for a given carbon chain. For example, consider propane (an alkane) and propene (an alkene). Both have 3 carbon atoms but propane is C3H8, whereas propene is C3H6. That is because propene contains a carbon-carbon double bond.
Propene is known as an unsaturated hydrocarbon because you could fit another two hydrogens around the carbon chain if you replaced the double carbon-carbon bond by a single one. The first 6 alkanes are:
Structural isomerism Alkanes from butane on have structural isomers. For example, in butane you can arrange the 4 carbon atoms and 10 hydrogens in two different ways: CH3CH2CH2CH3 which is just called butane; and
which is called 2-methylpropane. | |||||||||||||
|
Note: You need to check your syllabus to find out how much, if anything, you need to know about structural isomerism. If you do need to know about it, you should make sure that you at least know about the examples on the page about isomerism. | |||||||||||||
|
Chemically the isomers of butane behave in exactly the same way. That is because they both contain only carbon-carbon bonds and carbon-hydrogen bonds. The reactions that organic compounds undergo is determined by what sorts of bond are present. Combustion of alkanes Alkanes burn in air or oxygen. What you get depends on how much oxygen is available. Complete combustion In a plentiful supply of air, they burn to form carbon dioxide and water, and you get lots of heat evolved. For example, with methane or butane: CH4 + 2O2 2C4H10 + 13O2 | |||||||||||||
|
Note: You could be asked for the equation for almost any given alkane, so there is no point in learning these equations. You have to be able to work them out. Equations involving alkanes with an odd number of carbons are much easier than those with even numbers. If you aren't sure how to handle these, read the equations page. | |||||||||||||
|
Incomplete combustion If there isn't enough oxygen, the hydrogen always gets what is available first. You will always get water, but you will also get either carbon monoxide or carbon (as soot) formed as well as carbon dioxide. Carbon monoxide is colourless, odourless and poisonous, and so potentially very dangerous. It interferes with the way oxygen is carried around the body. Carbon monoxide combines irreversibly with a molecule in the blood called haemoglobin. Normally oxygen bonds to the haemoglobin, and gets carried around the body in the blood stream to where it is needed. The carbon monoxide binding with the haemoglobin prevents that happening. Reactions of alkanes with chlorine or bromine A mixture of methane and chlorine reacts on exposure to sunlight. A substitution reaction happens. A substitution reaction is one in which an atom in a molecule is replaced by a different atom. In this case, a hydrogen atom in the methane is replaced by a chlorine atom. CH4 + Cl2 The organic product, CH3Cl, is called chloromethane and is a colourless gas at room temperature, and you also get steamy fumes of hydrogen chloride gas. Similarly a mixture of methane and bromine vapour forms bromomethane and hydrogen bromide on exposure to sunlight. Bromomethane is also a gas at room temperature, and hydrogen bromide also forms steamy fumes. In both cases, it is the UV in the sunlight which triggers the reaction by splitting the chlorine or bromine molecules into individual atoms. Ethane, CH3CH3, will behave similarly forming CH3CH2Cl or CH3CH2Br. Things are a bit more complicated if you do the similar reactions with propane (or any other bigger alkane). With propane, CH3CH2CH3, the hydrogen being replaced could either be in the middle of the chain or at the end. That means that you will get both of these two different molecules formed if you were using chlorine: Cl 2-chloropropane: CH3CH ClCH3 | |||||||||||||
|
Note: This is all a massive simplification! In fact, if you do these reactions it is impossible to stop at the stage where just one hydrogen is replaced. You get a mixture of products that, in the methane and chlorine case, would include CH3Cl, CH2Cl2, CHCl3 and CCl4 as well, of course, as HCl. This isn't a useful reaction! I suspect it is only introduced at this level so that you meet the term "substitution reaction". | |||||||||||||
© Jim Clark 2021 |
|||||||||||||