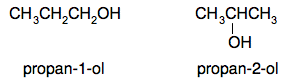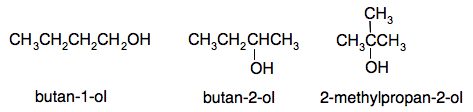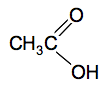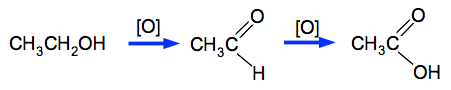|
Chemguide: Core Chemistry 14 - 16 Alcohols This page introduces alcohols - what they are, some simple examples, how they are made, and some simple properties. I am assuming that you have already read the pages about organic formulae, organic names and isomerism. There is a lot of chemistry on this page - don't rush it. What are alcohols? Alcohols are members of a homologous series (family) of molecules which have an OH group attached to a hydrocarbon chain. Their general formula is CnH2n+1OH. The substance normally called "alcohol" in everyday life is just one of the family and is properly called ethanol. The four smallest alcohols are:
Structural isomerism in the alcohols There is only one way in which you can arrange the atoms in methanol or ethanol to produce a molecule with an OH group, but both propanol and butanol have structural isomers containing OH groups. Structural isomerism in C3H7OH In C3H7OH, you could either put the OH group at the end of the chain or in the middle.
Notice the way that the position of the OH group is written in the name. Structural isomerism in C4H9OH In C4H9OH, you could again put the OH group at the end of the chain or in the middle, but there is a third isomer where the chain is branched.
| |||||||||
|
Note: If this isn't obvious to you, you must re-read the pages about organic names. and isomers | |||||||||
|
Physical properties of the alcohols Boiling points The boiling points of the alcohols are governed by the strength of the forces between their molecules (the intermolecular forces). | |||||||||
|
Note: If you haven't thought about intermolecular forces for a while (or at all!), you will need to read this page before you go on. | |||||||||
|
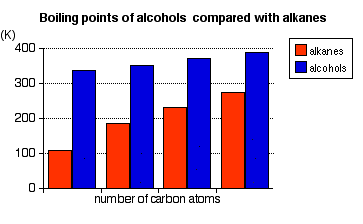
Alcohols have much higher boiling points than alkanes with the same number of carbon atoms. This diagram comes from an introduction to alcohols in the advanced part of Chemguide, and you might like to read the physical properties section of that page in more detail if you were interested. The level of knowledge you will need at the current level is only very superficial.
Notice that temperature is in Kelvin (K), not Celcius. O°C is 273 K. Intermolecular forces are much stronger in alcohols than in the corresponding alkane. Alkanes only have van der Waals dispersion forces. Alcohols also have dispersion forces, but in addition they have van der Waals dipole-dipole interactions and hydrogen bonds. The dipole-dipole attractions are because the oxygen is very electronegative, giving the molecules a permanent dipole. The hydrogen bonds arise from attraction between the slightly positive hydrogen attached to the oxygen and a lone pair on an oxygen atom on another molecule. Solubility in water The small alcohols are soluble in water, but solubility falls as the alcohol gets bigger. In order to get two liquids to mix, you have to break intermolecular forces in both liquids before anything else can happen. They will mix if new intermolecular forces can be set up between the two sorts of molecules which are strong enough to substitute for the original ones. Both water and alcohols have hydrogen bonding, and they can hydrogen bond with each other - so that works fine for small alcohols. However, as the chains of carbons in the alcohol get longer, they have to break hydrogen bonds between more and more water molecules in order to squeeze between them - but it is only the OH group in the alcohol which can form hydrogen bonds. With big alcohols, you aren't getting enough energy released when new intermolecular forces are set up to compensate for the energy needed to break the old ones. So solubility in water decreases as the alcohols get bigger. The manufacture of ethanol There are two different ways of doing this - by fermentation of carbohydrates using yeast, or by hydration of ethene. Making ethanol from carbohydrates by fermentation You are probably familiar with the use of yeast to make alcoholic drinks by fermenting carbohydrates of one sort or another. Alcohol can also be made for industrial use this way. Remember that alcohol is the common name for ethanol The starting material for the process varies widely, but will normally be some form of starchy plant material such as maize (US: corn), wheat, barley or potatoes. For simplicity, l am going to assume that we are going to get the ethanol from sucrose, C12H22O11 - ordinary sugar from sugar cane or sugar beet. The whole process is now a matter of biochemistry, and is governed by enzymes - biological catalysts. Important conditions
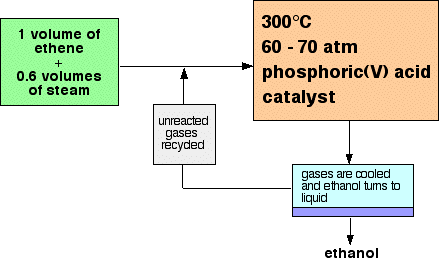
The reason that air has to be kept out is that ethanol can undergo oxidation in the air to form ethanoic acid - vinegar. The reactions These are summary descriptions and equations for a whole lot of separate small stages controlled by enzymes in the yeast. The sucrose is first split into two smaller sugars, glucose and fructose. These both have the formula C6H12O6, and so are structural isomers - molecules with the same molecular formula but a different arrangement of their atoms. C12H22O11 + H2O Enzymes in the yeast now break these down to give ethanol and carbon dioxide. C6H12O6 Separating the ethanol This can be done by fractional distillation of the reaction mixture. You will find fractional distillation of this mixture described by following this link. Industrially, this will simply be on a much bigger scale using metal rather than glass equipment. Making ethanol from ethene Ethanol is manufactured by reacting ethene with steam. The catalyst used is phosphoric(V) acid coated onto solid silicon dioxide as a support. The reaction is reversible. CH2=CH2 + H2O Only 5% of the ethene is converted into ethanol at each pass through the reactor. By removing the ethanol from the equilibrium mixture and recycling the ethene, it is possible to achieve an overall 95% conversion. A flow scheme for the reaction looks like this:
| |||||||||
|
Note: This is a bit of a simplification! When the gases from the reactor are cooled, then excess steam will condense as well as the ethanol. The ethanol will have to be separated from the water by fractional distillation. | |||||||||
|
You might wonder why the reaction mixture contains so little steam. The reason is that if you had too much steam, you risk diluting the catalyst, or even washing it off its silicon dioxide support. Advantages and disadvantages of using the two methods The main disadvantage of fermentation is that it is "batch" method. You have to make up a vat of sugar solution and yeast and then let it ferment for several days. This is slow and time consuming. You also also only get a fairly dilute solution of ethanol. Yeast is killed when the alcohol concentration exceeds about 15%. The main advantages of using fermentation are that it uses a renewable resource, and also uses very mild reaction conditions. The main advantage of making it from ethene is that it is a continuous flow process. You can take ethene directly from an oil refinery, react it with steam and produce ethanol in one continuous process. It is fast and efficient. Its main disadvantages are that it uses a non-renewable resource, and expensive high temperatures and pressures. Reactions of alcohols Alcohols burn All alcohols burn but the only alcohol you are ever likely to want to burn is ethanol which can be used as a fuel. Notice that it burns with a clean blue flame to give carbon dioxide and water. CH3CH2OH + 3O2 Alcohols react with sodium metal We will just take ethanol as typical. You probably remember the reaction of sodium with water. If not, this brief video will remind you. There is enough heat evolved to melt the sodium, and it rushes around the surface giving off hydrogen. An alkaline solution of sodium hydroxide is formed. 2Na + 2H2O Sodium behaves similarly, but much more slowly, if you add it to ethanol. This time the sodium sinks because it is denser than the ethanol, and hydrogen is again given off. An alkaline solution is formed, and if you add enough sodium you will eventually get the other product, sodium ethoxide, as a white solid. 2Na + 2CH3CH2OH The video shows the effect of adding a lot of sodium to some ethanol and ending up with solid sodium ethoxide. The two cases are very similar because what you are doing in each case is essentially the same. In water, HO-H, one of the hydrogens is replaced by sodium to give sodium hydroxide. You could write this as HONa. (Although you don't. In inorganic chemistry, the metal is usually written first.) With ethanol, CH3CH2O-H, the hydrogen in the OH group is again replaced to give CH3CH2ONa. (In organic chemistry, the organic bit is usually written first - although when you name it, the metal is named first. Confusing? Yes, but it is something you soon get used to.) If you draw a structural formula which shows the bonding around the sodium end, be very careful! The bonding between sodium and the rest of the formula is ionic. So the structural formula should properly be CH3CH2O- Na+. In an exam, if you draw a line between the oxygen and the sodium (showing a covalent bond), you will lose the mark for that structure.The catalytic dehydration of ethanol Ethanol can be dehydrated to give ethene by passing ethanol vapour over hot aluminium oxide as a catalyst. CH3CH2OH You might have wondered what the sand was doing in the flask with the ethanol. It isn't taking any part in the reaction - just helping the ethanol to boil smoothly. In case you didn't notice it, the ethanol flask was being heated by an electric heater. Other bigger alcohols behave is a similar way provided there is at least one hydrogen attached to the carbon next door to the one with the OH group. To make a double bond, you need to remove the OH group and one hydrogen from the next carbon atom. Oxidation of ethanol Ethanol can be oxidised to ethanoic acid (old name acetic acid - found in vinegar). Ethanoic acid is a member of the homologous series of carboxylic acids which contain a COOH group. There are two oxidising agents which may be mentioned in syllabuses - acidified potassium dichromate(VI) solution and acidified potassium manganate(VII) solution. In each case they are acidified with dilute sulfuric acid.
The commonly used one is potassium dichromate(VI). Potassium manganate(VII) isn't a reagent of choice in organic chemistry because it is such a powerful oxidising agent that it tends to chew up organic molecules and not just do the reaction you want it to. If your syllabus specifically mentions potassium manganate(VII) you will have to go with it, otherwise just ignore it. The equations for these reactions are really difficult if you try to write them properly. So in organic chemistry we simplify things by writing [O] to stand for oxygen from an oxidising agent. That will be perfectly acceptable in an exam at this level. CH3CH2OH + 2[O] Notice the way the formula of ethanoic acid is written. The two oxygens are written separately because they are bonded differently.
In fact, this oxidation takes place in two stages:
The first oxygen removes 2 hydrogen atoms (producing water) and gives a compound called ethanal. This is a member of the homologous series called aldehydes. You do not need to know about these at this level. The second oxygen slips in between the carbon and hydrogen atom on the right-hand side. In order to go all the way to the acid, you need two important conditions.
This next video explains the whole process using acidified potassium dichromate(VI) solution to oxidise ethanol. It is aimed at advanced level chemistry, but all you need to do is to ignore the mention of the aldehyde. Summary To oxidise ethanol to ethanoic acid using acidified potassium dichromate(VI) solution:
If you are using potassium manganate(VII) solution, the only difference is that the colour change is from deep purple to colourless (although if you were using excess oxidising agent, you would never see it go colourless - just a lighter shade of purple!). | |||||||||
|
Note: To be strictly accurate, when you distil the solution you get both water and acid distilling over. We just accept this - it is quite difficult to separate them by distillation alone. | |||||||||
|
Oxidation of other alcohols This is more complicated because it depends on the position of the OH group on the chain. Check your syllabus to see whether you need to know any of this - most syllabuses will probably restrict it to ethanol. If that's the case, ignore the rest of this. Cases just like ethanol This is where the OH group is at the end of the chain - for example, propan-1-ol CH3CH2CH2OH + 2[O] Propanoic acid is formed. Cases where the OH group isn't at the end of the chain Propan-2-ol is the simplest example.
You can see that after the first stage of the ethanol oxidation, there isn't anywhere else for it to go. If you compare this with the similar flow scheme above, you will see that there isn't room on the C=O carbon to add an OH group as well. You can't make an acid unless the OH group is on the end of the chain. Formation of esters from alcohols This is perhaps the most confusing part of organic chemistry at this level, and I am going to leave it to a page of its own. If you want to look at it now, you can follow this link to the page about esters, but I would suggest that you leave it until after you know some more about carboxylic acids. Uses of ethanol Ethanol has a wide range of uses, but most syllabuses only mention two. It is used as a solvent in all sorts of contexts, for example in the manufacture of cosmetics, drugs, detergents, and inks. It is also used as a fuel usually mixed in various proportions with petrol (gasoline) to give gasohol . The chart shows some of the combinations.
You do not need to know all this - I include it for interest only. The diagram comes from Wikipedia and was originally produced in 2011. Things have moved on a lot since then, and can't be taken as accurate now. There is controversy about the environmental advantages / disadvantages of using alcohol as a motor fuel. If you are interested, do some research to find the current thinking.
© Jim Clark 2021 |
|||||||||